Regulatory publishing professionals are adapting to AI-enabled workflows by developing new skills in data interpretation, AI literacy, and regulatory intelligence analysis.
Explore how agentic AI systems are improving regulatory submissions through autonomous workflows while maintaining essential human oversight and quality standards.
Regulatory operations teams are shifting from tactical, manual tasks toward strategic functions as AI platforms automate routine submission work.
Discover how microservices architecture on Azure eliminates regulatory publishing bottlenecks, reduces submission times by 50%, and ensures compliance scalability for pharmaceutical teams.
Transform regulatory operations with automated workflows connecting planning, document management, and eCTD publishing into seamless digital processes.
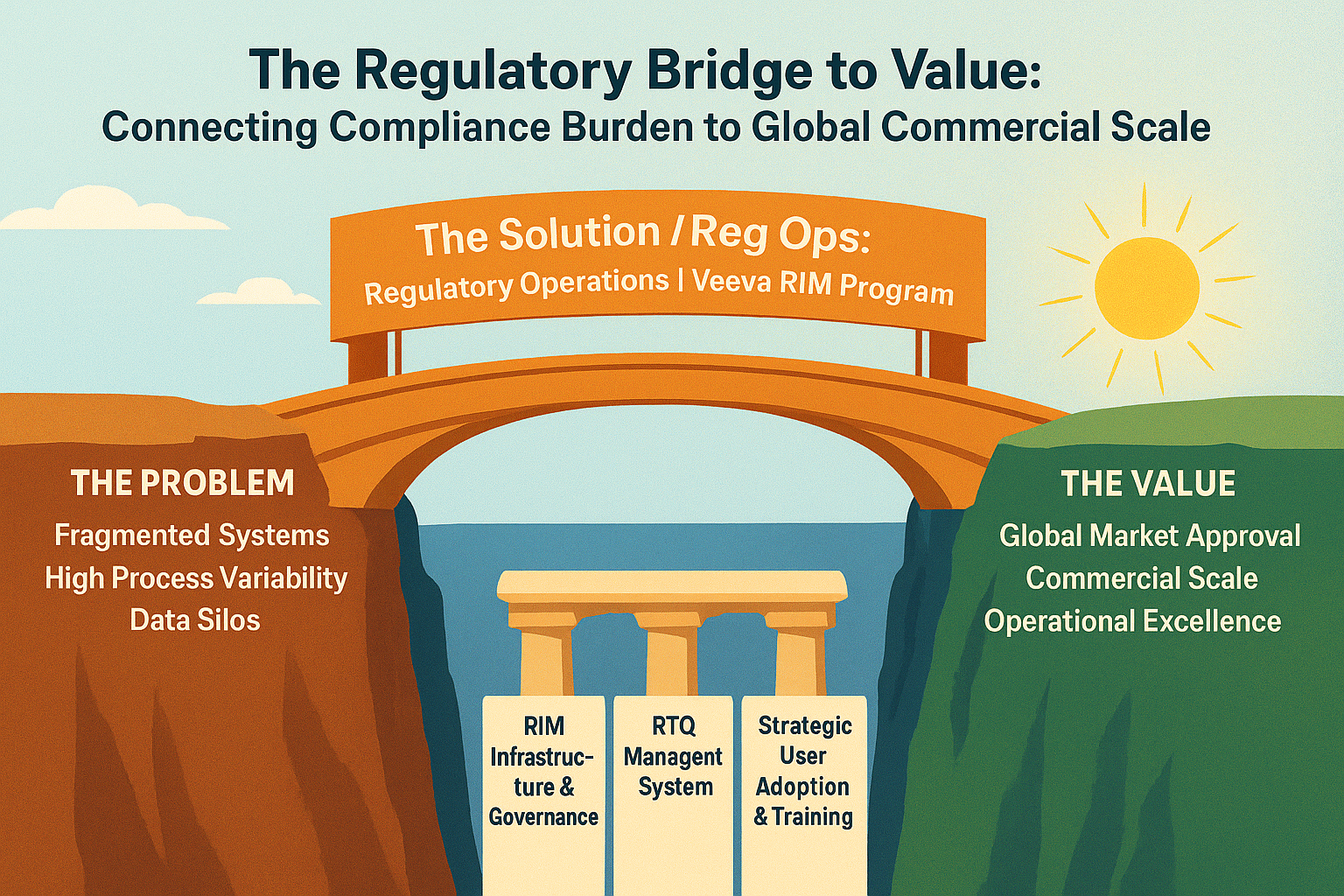
The regulatory landscape continues to evolve at an unprecedented pace. Between expanding global filing requirements, increasing agency expectations for electronic submissions, and the ongoing integration of AI and automation technologies, Directors of Regulatory Operations face a pivotal moment. The strategies and systems you put in place now will determine whether your organization leads or lags […]
As a Director of Regulatory Operations, you’ve likely built your document management processes around a system that once seemed adequate. But as submission volumes grow and regulatory requirements evolve, the cracks begin to show. Here are five warning signs that your current regulatory document management system is creating bottlenecks instead of enabling efficiency. 1. Your […]
Move beyond compliance to strategic regulatory operations. Scalability, data quality, and submission intelligence insights.
How AI is transforming regulatory affairs from document management to intelligent compliance. Enterprise authoring and analysis at scale.